RIGHTHEMICOLECTOMY AND EXTENDED RIGHT HEMICOLECTOMY
Cancer of the colon can spread to pericolic lymph nodes up to 5 to 6 cm from the tumour. Removal of at least 10 cm of bowel on either side of the tumour is recommended for clearance of these nodes.

The 10 cm of colon on either side of cancer of the ascending colon/ cecum is supplied by the ileocolic artery, the right colic artery, and the right branch of the middle colic artery. Ligation of these vessels entails removal of the terminal 10cm of ileum, cecum, ascending colon, hepatic flexure of the colon and the right part (usually one-third) of the transverse colon. For cancer of the hepatic flexure, an extended right hemicolectomy is done with additional removal of the transverse colon along with both branches of the middle colic artery.

VASCULAR VARIATIONS
The ileocolic artery:
The only constant branch of the SMA to the right half of the colon is the ileocolic artery. This constant structure is especially important for laparoscopic right colectomy, because the first step is grasping and dividing the ileocolic pedicle before mobilization of the mesocolon.
The right colic artery:
The right colic artery has been portrayed inaccurately in standard textbooks propagating misunderstandings of the true normal pattern. The right colic artery is not a constant branch of the SMA.
The right colic artery is an uncommon branch (10.7 percent) of the SMA. Blood supply to the ascending colon is mostly from a branch emanating from the ileocolic artery (66 percent) or the middle colic artery (23.3 percent).


The ileocolic vein:
The ileocolic vein (ICV), like the ileocolic artery (ICA), is a constant structure. There is only one vein that invariably joins the SMV.
The middle colic vein:
The middle colic vein accompanies the middle colic artery and usually drains into the SMV.
The right colic vein:
The right colic vein (RCV) is an inconstant vein that accompanies the right colic artery when present.
The superior right colic vein:
Superior right colic vein (SRCV) is the more constant right colic veins. It is the tributary from the marginal veins of the hepatic flexure (the RCV drains the blood from the marginal veins of the ascending colon). It always drains into the gastrocolic trunk (GCT).


The SRCV is the main cause of bleeding by avulsion during right hemicolectomy. It has no corresponding artery. It is the main cause of tension in the extraction of the surgical specimen after right hemicolectomy.
The SRCV should be ligated next to the gastrocolic trunk of Henle. Its ligation proximal to the GCTH increases the length of the transverse colon exteriorization for anastomosis, by about 3 cm.
The Gastrocolic Trunk Of Henle:
The anatomy of the GCT is complex. It is short (less than 25 mm) but with a major calibre (3–10 mm). Inadvertent traction by the first assistant is a well-known cause of severance of the fragile veins draining into it.
The gastrocolic trunk of Henle(GCT) is formed commonly by the union of three veins:
• The anterior superior pancreaticoduodenal vein (ASPDV),
• The right gastroepiploic vein (RGEV) and
• The superior right colic vein (SRCV)

Apart from the SRCV, many other colic veins might drain into the GCT.
• The right colic vein (RCV), which accompanies the RCA when present, commonly drains into the GCT (84%).
• The middle colic vein (MCV) uncommonly drains into the GTH (12%).
• When there are multiple accessory MCVs (aMCV), they may drain into the GTH.
For right hemicolectomy, careful identification and division of these single or multiple right colic veins draining into the GTH is required.


Crossing patterns of the arteries:
The SMA runs lateral and dorsal to the SMV as they enter the small bowel mesentery. Consequently, the ICA and the RCA can run anterior or posterior to the SMV.
Ligation of these arteries at the origin will be a surgical challenge, if they run posterior to the SMV. Recognizing this pattern in preoperative imaging will help in planning the approach to the origin of these vessels. Operating under the SMV can be hazardous, particularly laparoscopically.
The RCA crossing behind the SMV is likely to pose the most difficult challenge. The ICA can be ligated by flipping the small bowel loops to the upper left quadrant bringing the dorsal aspect of the SMV into perspective.
The ICA and RCA are commonly ligated on the left border of the SMV, if they run posterior to SMV, especially in an obese patient where the dissection on th edorsal surface of the SMV might be too risky .

FUSION FASCIA OF THE RIGHT MESOCOLON:
Fascia of Toldt:
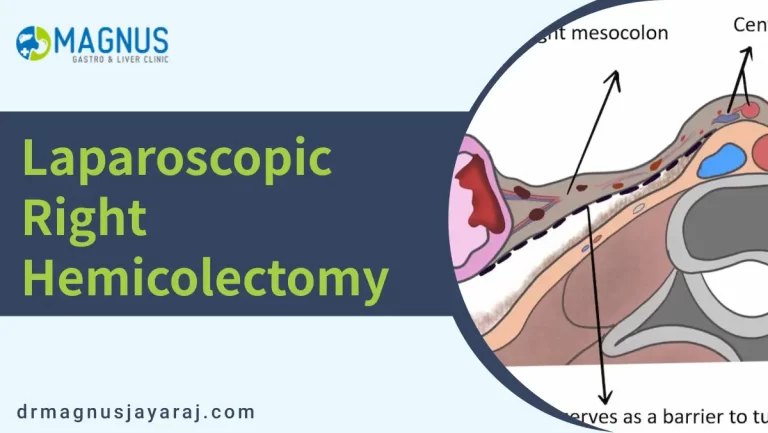
Traditionally, the right colon has been described as a retroperitoneal structure. At the end of the intestinal rotation, the mesentery of the right colon fuses with the parietal fascia of the retroperitoneum to form the fusion fascia of Toldt’s.
However, there is a potential space(Toldt’s space or retro-mesocolic space), filled with loose connective tissue such as angel’s hair, between the parietal fascia of the retroperitoneum and the visceral fascia of the right mesocolon.
Dissection in this avascular space during right hemicolectomy avoids injuries to the retroperitoneal structures(like the right ureter and the gonadal vessels).

Similarly, when the duodenal C loop and the pancreas become retroperitoneal after intestinal rotation, two fusion fascia form:
Fusion fascia of Treitz: adhesion plane between the visceral peritoneum of the duodenum and pancreas and the retroperitoneum.
Fusion fascia of Fredet: adhesion plane between visceral peritoneum of the mesocolon of ascending colon and hepatic colonic flexure and the visceral peritoneum of duodenum and pancreas.

As the dissection in the Toldt’s space proceeds cranially, theswitching membrane is divided to enter the fusion fascia of Fredet on the anterior surface of thepancreaticoduodenal complex. The medial limit of this fascia is the junction ofthe GCT with the SMV.
D3 LYMPHADENECTOMY
The Japanese Research Society for Cancer of the Colon and Rectum classification of colonic lymph nodes describes three levels of nodes.

•Paracolic LNs – nos. 201,202, and 203.
•Intermediate LNs include nos. 211, 212, and 213.
•Principle LNs include nos. 221, 222, and 223.

Corresponding to these three levels, there are three types of dissection beds: D1, D2 or D3, depending on the level of vessel dissection.
Standard right hemicolectomy usually removes up to the intermediate nodes and is considered a D2 dissection.
D3 lymph node dissection is recommended by Japanese guidelines for all colonic tumors invading the muscularis propria or deeper. It requires ligation of the arteries at their origin.

TheD3 area has been described well:
• the cranial border – running 5 mm proximal to the line connecting the origins of the GTH and the MCA;
• the medial border – running along the left-hand side of the SMA;
• the caudal border – running 5 mm distal to the line connecting the origin of the ICA; and
• the lateral border – running 1 cm along the right-hand side of the SMV.
D3 lymph node dissection contributes not only to the local control of disease but also, to accurate staging.

COMPLETE MESOCOLIC EXCISION
With the understanding that the colon is not a real retroperitoneal structure, anatomic studies have defined the mesocolon of the right colon, covered by mesocolic fascia dorsally and ventrally. All the lymphatics from the colon run within this envelop to the nodes at the origin of the arteries supplying that part of the colon. Surgery in the mesocolic plane forms an intact peritoneal and fascial-lined package of mesentery containing all potential routes of metastatic tumour spread. Dissection in this space also avoids injury to the retroperitoneal structures including the ureter and the gonadal vessels which are covered by the parietal fascia.


The concept and procedure for complete mesocolic excision (CME) was initially proposed by Hohenberger et al. in 2009. Removal of the colonic tumor with the complete mesocolon whose envelope is intact is expected to result in better tumor clearance and improved survival.
CME has two components:
• The visceral fascia is dissected sharply from the retroperitoneal fascia. Any breach of the visceral fascia layer, which potentially may lead to tumour spread within the peritoneal cavity, is avoided.
• Central vascular ligation (CVL)- the arteries to the tumor are ligated at their origin.
Pros:
•CME is a more oncologically sound procedure than a standard colonic resection as it includes a more radical lymph node dissection within an intact fascial envelope to reduce risk of tumor spread.
•Higher numbers of lymph nodes more accurately stage a patient and may improve survival in its own right.
Cons:
•Being a longer and more technically challenging operation, a higher complication rate is expected.
•The possibility of occurrence of catastrophic complications that might occur less frequently in a standard procedure.
•Oncological benefit has not been proven.
STANDARD VERSUS EXTENDED LYMPHADENECTOMY
“Highligation,” “central vascular ligation,” “complete mesocolic excision,” and “D3resection” are terms used to describe extended lymphadenectomy, beyond the primary feeding vessel and associated central (D2) lymph node basin.
According to the American Society of Colon and Rectal Surgeons Clinical Practice Guidelines for the Treatment of Colon Cancer, routine performance of extended lymphadenectomy is not recommended. Grade of Recommendation: Strong recommendation based on moderate-quality evidence, 1B.

Lymph node metastasis outside the standard field of resection (ie, proximal to primary feeding vessel and associated central (D2) nodes) occurs in 3% to 11% of colon cancers and is more likely with advanced T-stage cancers. Central lymph node involvement in the absence of pericolic or intermediate lymph node involvement (“skip metastases”) occurs in≤2% of cases.
Although routine performance of extended lymphadenectomy is not supported by the data available, dissection and retrieval, or at minimum, biopsy of clinically positive or suspicious lymph nodes outside the standard field of resection is recommended.
SMA vs SMV AS LANDMARKS FOR EXTENDED RESECTIONS
According to the mesenteric lymph node drainage rules and the Japanese Classification of Colorectal Carcinoma guidelines, radical surgery of right-sided colon cancer requires D3 cleaning of the lymph nodes at the root of the colonic vessels, requiring exposure of the superior mesenteric artery (SMA) and dissection of its right regional lymph nodes.
However, cleaning of the root nodes of SMA can result in potential complications such as severe diarrhea and chylous fistula. A study from Japan showed that the lymphatic drainage in the right-sided colon rarely drained to the left side of the anterior surface of the SMV.

Because of the difficulty in clearing the root nodes of SMA, most surgeons locate the internal boundary of D3 cleaning to the SMV. In the study of Gillot et al., the term ‘surgical trunk’ was used to describe the adipose tissue between the ileocolic vein caudally and Henle’s trunk (gastrocolic trunk) cranially. Lymph node dissection along this surgical trunk, meaning only the rightventrolateral SMV, is used for central lymph node dissection instead of dissection to the root of the main artery.
MEDIAL TO LATERAL APPROACH
Conventionally, during standard hemicolectomy in open surgery, the right colon is mobilised from the right lateral wall as the first step (lateral to medial approach). The lateral-to-medial dissection when used in laparoscopic colectomy is technically demanding due to limited operative space and insufficient manoeuvrability of the straight laparoscopic forceps.

In laparoscopic surgery, a medial to lateral approach is most commonly used as it has been found to be advantageous.
Pros of the medial to lateral approach:
•Attachment of colon to lateral abdominal wall exerts counter traction to the uplifted mesocolon and aids the initial mesenteric dissection. In contrast, early mobilization of colon in the lateral approach increases redundancy and hinders subsequent dissection.
•Early dentification of the ureter and gonadal vessels reduces the risk of damage.
•Early division of vascular pedicles reduces subsequent bleeding from dissection.
•It has the advantage of abiding by the ‘no touch’ principle for oncologic surgery. The vessels and mesenteric division prior to tumor-containing bowel mobilization could minimize cancer cell spread though the draining vessels.
•This approach along the vascular anatomy (superior mesenteric vessels) could lead to a more complete D3 lymphadenectomy.
An EAES consensus statement recommended that the medial-to-lateral technique should be the preferred approach for laparoscopic colon dissection. Rarely, when the medial mobilisation is difficult due to adhesions in the retroperitoneum, a lateral or a combined approach is used.

POSITIONS AND PORTS
Patients are most commonly placed supine with the surgical team located on the patient’s left side, where the surgeon stands in the middle, the scrub nurse to the surgeon’s left, and the camera assistant to the surgeon’s right. Another option is the use of the so-called French positioning, where the patient is placed in supine position with split legs, or alternatively, if a split-leg table is not available, the patient can be placed in low lithotomy position. With this setup, the surgeon stands in between the patient’s legs, the camera assistant to the surgeon’s right, and the scrub nurse to the right of the camera assistant.

There are several options for trocar positioning for laparoscopic right hemicolectomy. Generally, there are no firm rules for port placement beyond those of triangulating based on pathology/right lower quadrant. Over time, the surgeon will eventually identify their preferred configuration and alternatives when required based on body habitus, adhesions from prior surgery, and need for additional assist trocars.

The camera port location is placed at the apex of the pneumoperitoneum; most commonly, a 10mm camera port is placed in the supraumbilical position, which will also function as the site of exteriorization and extraction of the specimen. A 12mm port is used for the stapler. Additional 5mm working ports are placed (usually 2-3), according to the surgeon’s preference and position.
The inferior ports should be a minimum of two fingerbreadths lateral and superior to the anterior superior iliac spine to prevent limitation of movement by the patient.
ENTERING TOLDT’S SPACE
The ileocolic pedicle is used as the landmark to start the surgical procedure. It is identified by using atraumatic graspers to elevate the ileocecal junction toward the right lower quadrant and anteriorly toward the abdominal wall, placing the mesentery under tension.
The ileocolic pedicle can be seen to tent the peritoneum like a “bow string”. Incising the peritoneum beneath this pedicle allows entry into Toldt’s space. The junction between the ileocolic and the superior mesenteric vessels is identified as a V shaped configuration, in order to avoid inadvertent incision of the mesentery on the left side of the SMV which can result in loss of orientation and injury of the SMV and SMA.



Alternately, the patient is placed in the Trendelenburg position at 15 to 30° and the body titled to the left by 15° to facilitate the small intestine falling on the left side of the abdominal cavity. The terminal ileum is lifted to separate the small bowel mesentery from the retroperitoneum, anterior to the right iliac artery, to reach the Toldt’s space.


THE RETROMESOCOLIC TUNNEL
The dissection is usually very quick, mostly blunt, and greatly aided by CO2 insufflation. The only retraction needed is the upward elevation of the mesentery. The surgeon’s left hand is used to support the ileocolic pedicle, while the right hand gently sweeps the retroperitoneal tissue down, away from the mesentery. It is quite easy to remain in the proper plane by following the fine areolar tissue, which connects the mesocolon to the retroperitoneum. Care should be taken not to broach Gerota’s fascia posteriorly, which encloses and protects the right kidney and ureter.

The dissection is continued laterally under the colon, but the white line of Toldt (lateral peritoneal attachment of the right colon) is left intact and the tumour is not touched until after the vascular pedicle is ligated. In the medial aspect, the duodenum should be clearly seen and preserved.
The retromesenteric dissection is continued cephalad, a plane below the transverse colon is realized when a purple/dark space is found deep to the transverse colon. This represents the space below the liver, into the lesser sac. This dissection effectively creates a blind-ending retroperitoneal tunnel below the right mesocolon.

Medially, dissection proceed anterior to the fascia of Fredet (the anterior pancreaticoduodenal fascia). The omental bursa is entered anterior to the duodenum and pancreas (anterior to the fascia of Fredet). Dissection in the area of the duodenum and the pancreas should be exceedingly gentle to prevent bleeding, pancreatitis, and injury.
PEDICLES LIGATION
The cephalad dissection is eventually limited by the ileocolic pedicle. At this point, the pedicle needs to be divided. Complete dissection of the second part of the duodenum off the ascending colon mesentery usually leads to thinning of that mesentery on both sides of the ileocolic vessels, such that an appropriate window can be readily made. The ileocolic vessels can then be isolated, secured between clips and divided near their origins. The ileocolic vein is better managed before the ileocolic artery because it is more prone to tearing.


Further dissection proceeds cranially along the anterior aspect of the SMV. The mesocolon is dissected off in a “page-turning” manner. The inferior part of the duodenum and then the anterior surface of the pancreas are exposed. In a minority of cases, a true right colic artery, originating from the superior mesenteric artery (SMA),is identified and similarly divided, divided after having assessed whether the right colic artery crosses anteriorly or posteriorly to the SMV. Further central dissection will lead toward the gastrocolic trunk, which is the upper limit of this part of the dissection.
The transverse colon is hoisted anteriorly to visualise the vessels in the transverse mesocolon. The SRCV is ligated close to its junction with the GCT. The middle colic vessels are identified by gentle elevation of the transverse colon off the duodenum and retroperitoneum. In cecal cancer, the right branches of the middle colic vessels are divided. For more distal tumours of the ascending colon or hepatic flexure, the entire MCA may be transected at its origin from the superior mesenteric artery (extended right hemicolectomy).

TRANSECTION OF THE BOWEL
The mesenteric transection is carried towards the chosen proximal margin in the distal ileum and also towards the chosen distal resection margin in the colon. The terminal ileum (10cm from the IC junction) and the transverse colon (junction of right 1/3 and left 2/3) are divided.

LATERAL MOBILISATION
The entire mesocolon is now fully mobilised and the specimen remains fixed only by the inferior and lateral attachments. The right lateral peritoneal reflection alongside the cecum and ascending colon is identified and scored. Lateral mobilization starts around the cecum and the appendix and then gradually the lateral suspension of the ascending colon is taken down.

CRANIAL FIRST
The anatomy of the region at the inferior border of the head and neck of the pancreas which is around 0.5–2.0 cm from the right side of the SMV is one of the most challenging aspects of pancreatic and right colonic resections for two reasons, namely, the fusion between the greater omentum and the transverse mesocolon at this level and the complex anatomy of the vulnerable venous tributaries of the SMV.

Fusion of the transverse mesocolon with the greater omentum and pancreas results in right gastroepiploic vein derived from the foregut and the SRCV derived from the terminal part of the midgut draining through a common trunk (the GCT) into the SMV. GCT and its branches have close anatomic relations to right colon arteries as well: the most common course of these arteries is caudal and parallel.

The GCT and its branches are vulnerable to injury and haemorrhage during right hemicolectomy, and hence are dissected first in the cranial- first approach. The gastrocolic trunk of the Henle region is easier to reach with the cranial approach after dividing the gastrocolic ligament, entering the lesser sac, recognizing the fold of the right gastroepiploic vessels and then proceeding to the right side towards the hepatic flexure along embryological planes.

The omental bursa is first opened wide, and the gastrocolic trunk of Henle is exposed, using the right gastroepiploic vessels and the accessory right colic vein (ARCV) as landmarks. The transverse mesocolon is detached from the inferior border of the pancreatic neck, revealing the superior mesenteric vein(SMV). The anterior aspect of the gastro colic trunk is dissected and the colic veins that enter it anteriorly are divided.



Further dissection can proceed on the SMV downwards in the Cranio-caudal approach. Alternately, in the combined approach, a gauge piece is placed at the origin of the GCT on the SMV- this area is approached from the ileocolic pedicle dissecting along the anterior surface of the SMV as described previously.
ARTERY FIRST
When the objective is to clear the nodes at the roots of the arteries, the SMA becomes the landmark for dissection instead of the SMV.
The mesocolon is dissected between the superficial layer of the fat tissue and the deep layer of the vascular sheath along the superior mesenteric artery. After the course of each branch is exposed, each supplying or draining vessel is transected at its root. Dissection close to the SMA may result in severe diarrhoea and there is a danger of damage to splanchnic nerves if the surgeon strips the root of the artery.


ANASTOMOSIS
Anastomotic technique (intracorporeal or extracorporeal) depends on the experience and choice of the surgeon.
For extracorporeal anastomosis, a mini vertical incision (4–6 cm depending on the bulkiness of the specimen and the thickness of the abdominal wall) at the level of the umbilicus is made by extending the supraumbilical port cephalad.

A wound protector is inserted, and the mobilized colon and ileum are exteriorized. The surgeon must ensure that adequate mobilization is performed to allow the colon and small bowel to be exteriorized with ease. Care should be taken to maintain the proper orientation of the ileal and colonic limbs. There are multiple anastomosis techniques, and the choice usually depends on the preference of the surgeon or a standardized approach in the institution. A lateral-lateral anastomosis, either manual or mechanical, is usually recommended.
For intracorporeal anastomosis, a mini-Pfannenstiel incision is generally used for the extraction of the specimen. Again, the choice of anastomosis depends on the surgeon’s preference. The proximal ileal and the distal colonic transection points are cleared from fatty attachments and the mesentery is divided about 2cm towards the remaining bowel ends.
For a stapled anastomosis, the two ends are oriented either in a peristaltic or in an anti-peristaltic fashion. The antimesenteric surfaces may be approximated with a seromuscular suture. An enterotomy is made in each limb for insertion of the limbs of the stapler. The limbs are anastomosed in side-to-side fashion. The resulting enterotomy is closed with a running intracorporeal suture (3–0 Vicryl) or with application of another GIA stapler.

MESENTERIC DEFECT CLOSURE
The mesenteric defect is closed with a running absorbable suture, although a retrospective study has questioned the utility of this practice (there were only 4 mesenteric defect-related complications among 530 patients in which the defect was not closed).
REFERENCES
Açar, Halil İbrahim, AyhanCömert, Abdullah Avşar,Safa Çelik,and Mehmet AyhanKuzu.“Dynamic Article: Surgical Anatomical Planes for Complete Mesocolic Excision and Applied VascularAnatomy of the Right Colon.” Diseasesof the Colon and Rectum 57,no. 10 (October 2014): 1169–75. https://doi.org/10.1097/DCR.0000000000000128.
Alsabilah, J., W. R. Kim, and N. K. Kim.“Vascular Structures of the Right Colon: Incidence and Variations with TheirClinical Implications.” ScandinavianJournal of Surgery: SJS: Official Organ for the Finnish Surgical Society andthe Scandinavian Surgical Society106, no. 2 (June 2017): 107–15. https://doi.org/10.1177/1457496916650999.
Bates,David D. B., Viktoriya Paroder, Chandana Lall,Neeraj Lalwani, Maria Widmar, and Julio Garcia-Aguilar.“Complete Mesocolic Excision and Central VascularLigation for Right Colon Cancer: An Introduction for Abdominal Radiologists.” AbdominalRadiology (New York) 44,no. 11 (November 2019): 3518–26. https://doi.org/10.1007/s00261-019-02037-9.
Benz,Stefan, Yu Tam, Andrea Tannapfel, and Ingo Stricker. “The UncinateProcess First Approach: A Novel Technique for Laparoscopic Right Hemicolectomywith Complete Mesocolic Excision.” SurgicalEndoscopy 30, no. 5 (May 2016): 1930–37. https://doi.org/10.1007/s00464-015-4417-1.
Bergamaschi,Roberto, Elie Schochet, Christopher Haughn, Marshall Burke, James F. Reed,and Jean-Pierre Arnaud. “Standardized Laparoscopic Intracorporeal RightColectomy for Cancer: Short-Term Outcome in 111 Unselected Patients.” Diseasesof the Colon and Rectum 51,no. 9 (September 2008): 1350–55. https://doi.org/10.1007/s10350-008-9341-1.
Chow,Carina F. K., and Seon Hahn Kim. “Laparoscopic Complete Mesocolic Excision: West Meets East.” WorldJournal of Gastroenterology20, no. 39 (October 21, 2014): 14301–7. https://doi.org/10.3748/wjg.v20.i39.14301.
Deng,Xiangbing, Tao Hu, Mingtian Wei, Qingbin Wu, Tinghan Yang, Wenjian Meng, and Ziqiang Wang. “Feasibility of aUnidirectionally Progressive, Pancreas-Oriented Procedure for Laparoscopic D3Right Hemicolectomy.” Langenbeck’sArchives of Surgery 403,no. 6 (September 2018): 761–68. https://doi.org/10.1007/s00423-018-1703-0.
Du, Songtao, Bomiao Zhang, Yanlong Liu, Peng Han, Chengxin Song, Fangjie Hu, Tianyi Xia, Xiangxin Wu, and Binbin Cui. “A Novel and Safe Approach:Middle Cranial Approach for Laparoscopic Right Hemicolon Cancer Surgery with Complete Mesocolic Excision.” SurgicalEndoscopy 32, no. 5 (May 2018): 2567–74. https://doi.org/10.1007/s00464-017-5982-2.
Feng,Bo, Jing Sun, Tian-Long Ling, Ai-Guo Lu, Ming-Liang Wang, Xue-YuChen, Jun-Jun Ma, et al. “Laparoscopic Complete Mesocolic Excision (CME) with Medial Accessfor Right-Hemi Colon Cancer: Feasibility and Technical Strategies.” SurgicalEndoscopy 26, no. 12 (December 2012):3669–75. https://doi.org/10.1007/s00464-012-2435-9.
Gao,Zhidong, Yingjiang Ye, Weiguang Zhang, Danhua Shen, Yanfeng Zhong, KeweiJiang, Xiaodong Yang, et al. “An Anatomical,Histopathological, and Molecular Biological Function Study of the Fascias Posterior to the Interperitoneal Colon and Its AssociatedMesocolon: Their Relevance to Colonic Surgery.” Journal of Anatomy 223, no. 2 (August 2013): 123–32. https://doi.org/10.1111/joa.12066.
He, Zirui,Hao Su,Kai Ye, Yueming Sun, Yincong Guo, Quan Wang, Yong Li, et al.“Anatomical Characteristics and Classifications of Gastrocolic Trunk of Henlein Laparoscopic Right Colectomy: Preliminary Results of Multicenter Observational Study.” SurgicalEndoscopy 34, no. 10 (October 2020):4655–61. https://doi.org/10.1007/s00464-019-07247-2.
He, Zirui,Sen Zhang, Pei Xue, Xialin Yan, LeqiZhou, Jianwen Li, Mingliang Wang, et al. “Completely MedialAccess by Page-Turning Approach for Laparoscopic Right Hemi-Colectomy:6-Year-Experience in Single Center.” SurgicalEndoscopy 33, no. 3 (March 2019): 959–65. https://doi.org/10.1007/s00464-018-6525-1.
Hida, Jin-ichi, Kiyotaka Okuno, Masayuki Yasutomi, Takehito Yoshifuji, Toshihiro Uchida, TadaoTokoro, and Hitoshi Shiozaki. “Optimal Ligation Level of thePrimary Feeding Artery and Bowel Resection Margin in Colon Cancer Surgery: TheInfluence of the Site of the Primary Feeding Artery.” Diseasesof the Colon and Rectum 48,no. 12 (December 2005): 2232–37. https://doi.org/10.1007/s10350-005-0161-2.
Hohenberger, W., K. Weber, K. Matzel, T. Papadopoulos, and S. Merkel.“Standardized Surgery for Colonic Cancer: Complete Mesocolic Excision and CentralLigation–Technical Notes and Outcome.” ColorectalDisease: The Official Journal of the Association of Coloproctology of GreatBritain and Ireland 11,no. 4 (May 2009): 354–64; discussion 364-365. https://doi.org/10.1111/j.1463-1318.2008.01735.x.
Ignjatovic, D., S. Sund,B. Stimec, and R. Bergamaschi. “VascularRelationships in Right Colectomy for Cancer: Clinical Implications.” Techniquesin Coloproctology 11,no. 3 (September 2007): 247–50. https://doi.org/10.1007/s10151-007-0359-5.
Ignjatovic, Dejan, Milan Spasojevic, and Bojan Stimec. “Can the Gastrocolic Trunk ofHenle Serve as an Anatomical Landmark in Laparoscopic Right Colectomy? A Postmortem Anatomical Study.” AmericanJournal of Surgery 199,no. 2 (February 2010): 249–54. https://doi.org/10.1016/j.amjsurg.2009.03.010.
Jin, Gang, Hongfang Tuo, Masanori Sugiyama, Atsuko Oki, Nobutsugu Abe, Toshiyuki Mori, Tadahiko Masaki, and Yutaka Atomi.“Anatomic Study of the Superior Right Colic Vein: Its Relevance to Pancreaticand Colonic Surgery.” AmericanJournal of Surgery 191,no. 1 (January 2006): 100–103. https://doi.org/10.1016/j.amjsurg.2005.10.009.
Kanemitsu, Yukihide, Koji Komori, Kenya Kimura, andTomoyuki Kato. “D3 Lymph Node Dissection in Right Hemicolectomy with a No-TouchIsolation Technique in Patients With Colon Cancer.” Diseasesof the Colon and Rectum 56,no. 7 (July 2013): 815–24. https://doi.org/10.1097/DCR.0b013e3182919093.
Lee, SeongDae,and Seok-Byung Lim. “D3 Lymphadenectomy Using a Medial to Lateral Approach forCurable Right-Sided Colon Cancer.” InternationalJournal of Colorectal Disease24, no. 3 (March 2009): 295–300. https://doi.org/10.1007/s00384-008-0597-7.
Levard, H., C. Denet,and B. Gayet.“Laparoscopic Right Colectomy from Top to Bottom.” Journalof Visceral Surgery 149,no. 1 (February 2012): e34-37. https://doi.org/10.1016/j.jviscsurg.2011.12.005.
Liang,Jin-Tung,John Huang, Tzu-Chun Chen, and Ji-Shiang Hung. “The ToldtFascia: A Historic Review and Surgical Implications in Complete Mesocolic Excision for Colon Cancer.” AsianJournal of Surgery 42,no. 1 (January 1, 2019): 1–5. https://doi.org/10.1016/j.asjsur.2018.11.006.
Matsuda,Takeru, Takeshi Iwasaki, Masaaki Mitsutsuji, Kenro Hirata, Yoko Maekawa, TomokoTanaka, Etsuji Shimada, and Yoshihiro Kakeji. “Cranial-to-Caudal Approach forRadical Lymph Node Dissection along the Surgical Trunk in Laparoscopic RightHemicolectomy.” SurgicalEndoscopy 29, no. 4 (April 2015): 1001. https://doi.org/10.1007/s00464-014-3761-x.
Matsuda,Takeru, Takeshi Iwasaki, Masaaki Mitsutsuji, Kenro Hirata, Yoko Maekawa, Daisuke Tsugawa, Yutaka Sugita, Yasuo Sumi, Etsuji Shimada, and Yoshihiro Kakeji. “Cranially Approached RadicalLymph Node Dissection around the Middle Colic Vessels in Laparoscopic ColonCancer Surgery.” Langenbeck’sArchives of Surgery 400,no. 1 (January 2015): 113–17. https://doi.org/10.1007/s00423-014-1250-2.
Matsuda,Takeru, Takeshi Iwasaki, Yasuo Sumi, Kimihiro Yamashita, Hiroshi Hasegawa,Masashi Yamamoto, Yoshiko Matsuda, et al. “Laparoscopic Complete Mesocolic Excision for Right-Sided ColonCancer Using a Cranial Approach: Anatomical and Embryological Consideration.” InternationalJournal of Colorectal Disease32, no. 1 (January 2017): 139–41. https://doi.org/10.1007/s00384-016-2673-8.
Mike,Makio,and Nobuyasu Kano. “Laparoscopic Surgery forColon Cancer: A Review of the Fascial Composition of the Abdominal Cavity.” SurgeryToday 45, no. 2 (February 2015): 129–39.https://doi.org/10.1007/s00595-014-0857-9.
Negoi, Ionut, Mircea Beuran, Sorin Hostiuc, Ruxandra Irina Negoi,and Yosuke Inoue. “Surgical Anatomy of the Superior Mesenteric Vessels Relatedto Colon and Pancreatic Surgery: A Systematic Review and Meta-Analysis.” ScientificReports 8, no. 1 (March 8, 2018): 4184. https://doi.org/10.1038/s41598-018-22641-x.
Ogino,Takayuki, Ichiro Takemasa, Genki Horitsugi, Mamoru Furuyashiki, Katsuya Ohta,Mamoru Uemura, Junichi Nishimura, et al.“Preoperative Evaluation of Venous Anatomy in Laparoscopic Complete Mesocolic Excision for Right Colon Cancer.” Annalsof Surgical Oncology 21 Suppl3 (June 2014): S429-435. https://doi.org/10.1245/s10434-014-3572-2.
Ogura,Atsushi, Ryutaro Kobayashi, Satoru Kawai, Kenji Takagi, Kiyotaka Kawai, Takashi Maeda, Tsukasa Aritake, Natsuki Nagano, and Satoaki Kamiya. “Cranial-First Approach ofLaparoscopic Left Colectomy for T4 Descending Colon Cancer Invading the Gerota’s Fascia.” SurgicalCase Reports 5,no. 1 (October 28, 2019): 159. https://doi.org/10.1186/s40792-019-0720-8.
Pellino, Gianluca, Oliver Warren, SarahMills, Shahnawaz Rasheed, Paris P. Tekkis, and Christos Kontovounisios. “Comparison of Western and AsianGuidelines Concerning the Management of Colon Cancer.” Diseasesof the Colon and Rectum 61,no. 2 (February 2018): 250–59. https://doi.org/10.1097/DCR.0000000000001012.
Shatari, T., M. Fujita, K. Nozawa, K.Haku, M. Niimi,Y. Ikeda, S. Kann, and S. Kodaira. “Vascular Anatomy for Right ColonLymphadenectomy.” Surgicaland Radiologic Anatomy: SRA25, no. 2 (May 2003): 86–88. https://doi.org/10.1007/s00276-003-0100-7.
Søndenaa, K., P. Quirke, W. Hohenberger, K. Sugihara, H. Kobayashi, H.Kessler, G. Brown, et al. “The Rationale behind Complete Mesocolic Excision (CME) and a CentralVascular Ligation for Colon Cancer in Open and Laparoscopic Surgery :Proceedings of a Consensus Conference.” InternationalJournal of Colorectal Disease29, no. 4 (April 2014): 419–28. https://doi.org/10.1007/s00384-013-1818-2.
Spasojevic, M., B. V. Stimec, J. F. Fasel,S. Terraz, and D. Ignjatovic. “3D Relations between Right ColonArteries and the Superior Mesenteric Vein: A Preliminary Study withMultidetector Computed Tomography.” SurgicalEndoscopy 25, no. 6 (June 2011): 1883–86. https://doi.org/10.1007/s00464-010-1480-5.
Spasojevic, Milan, Bojan V. Stimec, Anne Pernille H. Dyrbekk, Zvezdana Tepavcevic, Bjorn Edwin, Arne Bakka,and DejanIgnjatovic. “Lymph Node Distribution in theD3 Area of the Right Mesocolon: Implications for an Anatomically Correct CancerResection. A Postmortem Study.” Diseasesof the Colon and Rectum 56,no. 12 (December 2013): 1381–87. https://doi.org/10.1097/01.dcr.0000436279.18577.d3.
Strey, Christoph Werner, Christoph Wullstein, Michel Adamina, Ayman Agha, Heiko Aselmann, Thomas Becker, Robert Grützmann, et al. “Laparoscopic RightHemicolectomy with CME: Standardization Using the ‘Critical View’ Concept.” SurgicalEndoscopy 32, no. 12 (December 2018):5021–30. https://doi.org/10.1007/s00464-018-6267-0.
Tokuhara, Katsuji, Terufumi Yoshida, Yuki Matsui, KazuhikoYoshioka, and Mitsugu Sekimoto. “Laparoscopy-AssistedRight Hemicolectomy with the Bottom-to-Up Approach for Right-Side ColonCancer.” IndianJournal of Surgery,October 8, 2020. https://doi.org/10.1007/s12262-020-02618-9.
Tsai,Kai-Lung, Wei-Hung Lai, Ko-Chao Lee, Shung-Eing Lin, Chia-Lo Chang, Chien-ChangLu, Wan-Hsiang Hu, Seng-Kee Chuah, and Hong-Hwa Chen. “Long-TermConsequences of Nonclosure of Mesenteric Defects afterTraditional Right Colectomy.” BioMedResearch International 2018(2018): 9123912. https://doi.org/10.1155/2018/9123912.
Turati, L., G. Gallo, C. Zogno,A. Costanzo, E. Rausa, M. Trompetto, and G. Sgroi.“‘Up-to-down First’ Approach in Laparoscopic Right Colectomy with Complete Mesocolic Excision – Rationale and TechnicalNotes to Avoid Vascular and Pancreatic Lesions – a Video Vignette.” ColorectalDisease: The Official Journal of the Association of Coloproctology of GreatBritain and Ireland 21,no. 6 (June 2019): 730–31. https://doi.org/10.1111/codi.14630.
Uematsu, Dai, GakuAkiyama, and Akiko Magishi. “Multimedia Article. RadicalLymphadenectomy for Advanced Colon Cancer via Separation of the Mesocolon intoTwo Layers as in Filleting Fish.” SurgicalEndoscopy 25, no. 5 (May 2011): 1659–60. https://doi.org/10.1007/s00464-010-1439-6.
West,Nicholas P., Werner Hohenberger, Klaus Weber, Aristoteles Perrakis, Paul J. Finan,and Philip Quirke. “Complete Mesocolic Excision with Central VascularLigation Produces an Oncologically Superior Specimen Compared withStandard Surgery for Carcinoma of the Colon.” Journal of Clinical Oncology: OfficialJournal of the American Society of Clinical Oncology 28, no. 2 (January 10, 2010):272–78. https://doi.org/10.1200/JCO.2009.24.1448.
Wong,J. T., and M. A. Abbas. “Laparoscopic Right Hemicolectomy.” Techniquesin Coloproctology 17 Suppl1 (February 2013): S3-9. https://doi.org/10.1007/s10151-012-0931-5.
Wu, Chuying, Kai Ye, Yiyang Wu, Qiwei Chen, Jianhua Xu, Jianan Lin, and Wengui Kang. “Variations in Right ColicVascular Anatomy Observed during Laparoscopic Right Colectomy.” WorldJournal of Surgical Oncology17, no. 1 (January 12, 2019): 16. https://doi.org/10.1186/s12957-019-1561-4.
Yi,Xiao-Jiang, Xin-Quan Lu, Hong-Ming Li, Wei Wang, Wen-Jun Xiong,JinWan, and De-Chang Diao. “Feasibility and Efficacy ofLaparoscopic Radical Right Hemicolectomy with Complete Mesocolic Excision Using an ‘artery-First’Approach.” GastroenterologyReport 7, no. 3 (June 2019): 199–204. https://doi.org/10.1093/gastro/goy047.
Young-Fadok,T. M., and H. Nelson. “Laparoscopic Right Colectomy: Five-Step Procedure.” Diseasesof the Colon and Rectum 43,no. 2 (February 2000): 267–71; discussion 271-273. https://doi.org/10.1007/BF02236994.
Zenger,Serkan, ErmanAytac,Bulent Gurbuz, Volkan Ozben,Emre Ozoran, Bilgi Baca, Emre Balik,Ismail Hamzaoglu, Tayfun Karahasanoglu, and Dursun Bugra. “Metastasis to Lymph Nodes aroundthe Vascular Tie Worsens Long-Term Oncological Outcomes Following Complete Mesocolic Excision and ConventionalColectomy for Right-Sided Colon Cancer.” Techniquesin Coloproctology 25,no. 3 (March 2021): 309–17. https://doi.org/10.1007/s10151-020-02378-4.
Zhang,Ce, Zi-Hai Ding, Hai-Tao Yu, Jiang Yu, Ya-Nan Wang, Yan-Feng Hu, and Guo-XinLi. “Retrocolic Spaces: Anatomy of the SurgicalPlanes in Laparoscopic Right Hemicolectomy for Cancer.” TheAmerican Surgeon 77,no. 11 (November 2011): 1546–52.
Zhao,Li-Ying, Hao Liu, Ya-Nan Wang, Hai-Jun Deng, Qi Xue,and Guo-Xin Li. “Techniques and Feasibility of Laparoscopic Extended RightHemicolectomy with D3 Lymphadenectomy.” WorldJournal of Gastroenterology20, no. 30 (August 14, 2014): 10531–36. https://doi.org/10.3748/wjg.v20.i30.10531.
Zhu,Da-Jian, Xiao-Wu Chen, Man-Zhao OuYang, and Yan Lu. “Three SurgicalPlanes Identified in Laparoscopic Complete Mesocolic Excision for Right-Sided ColonCancer.” WorldJournal of Surgical Oncology14, no. 1 (January 12, 2016): 7. https://doi.org/10.1186/s12957-015-0758-4.
Read also best laparoscopic surgeon in India.